Introduction
Among the numerous types of post-translational protein modifications (PTMs), phosphorylation is a widespread class of modifications. Phosphorylated proteins account for approximately 1/3 to 2/3 of all eukaryotic proteins. Whereas, quantitative analysis of phosphorylation modification sites has been challenging because the extent of site-specific phosphorylation modifications may range from less than 1% to greater than 90% while often remaining low. Highly specific isolation enrichment methods can greatly improve the analysis depth of phosphorylated proteomes.
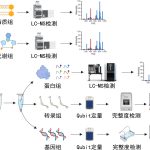
The DIA technology combined with highly specific phosphoproteome isolation techniques realizes high fidelity and high-depth protein phosphorylation modification profiling and analysis.
The 4D-protein phosphorylation modification omics analysis which combines 4D-proteomics technology with the existing protein phosphorylation modification omics analysis can obtain protein phosphorylation modification profiling with higher accuracy compared with conventional techniques and offers technical support for related biological and medical research.
Application Scenarios:
For phosphorylation proteomics analysis
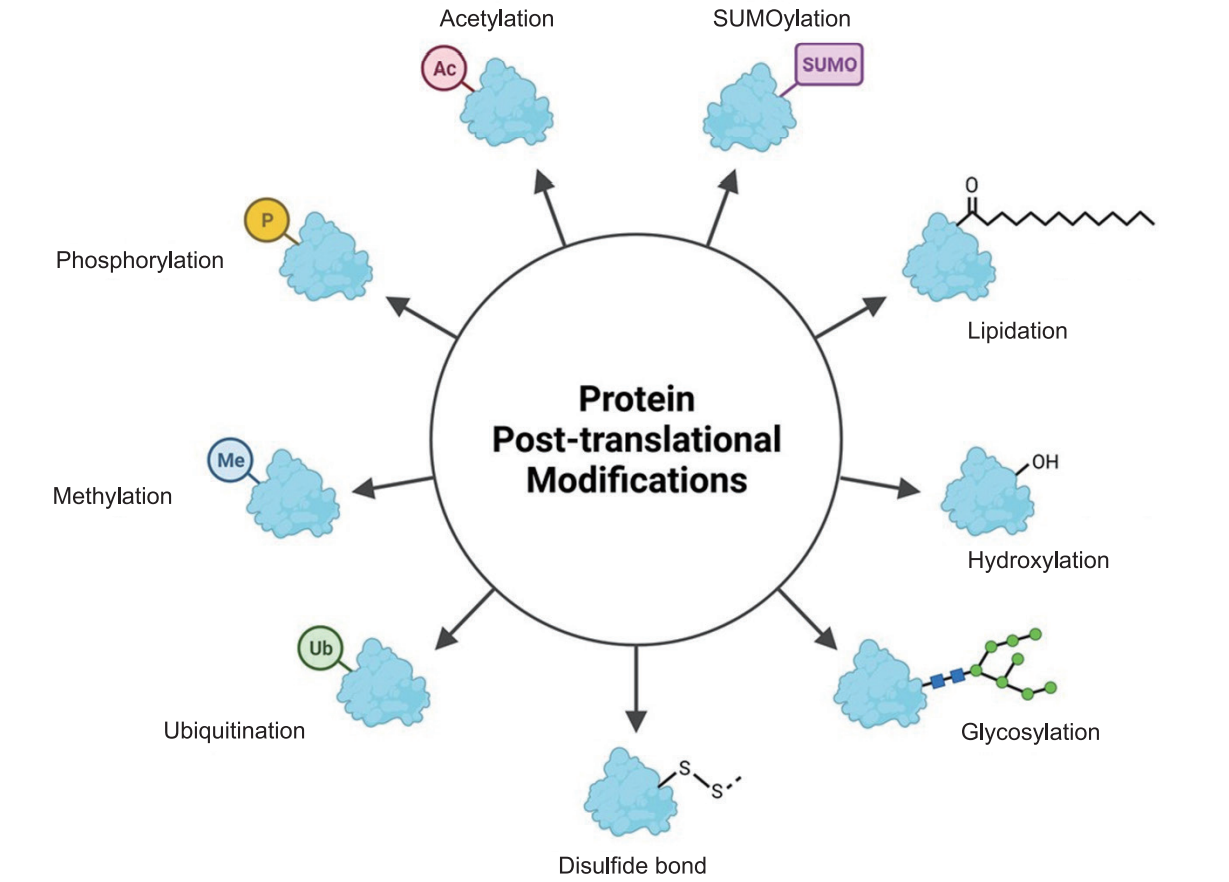
Common PTMs
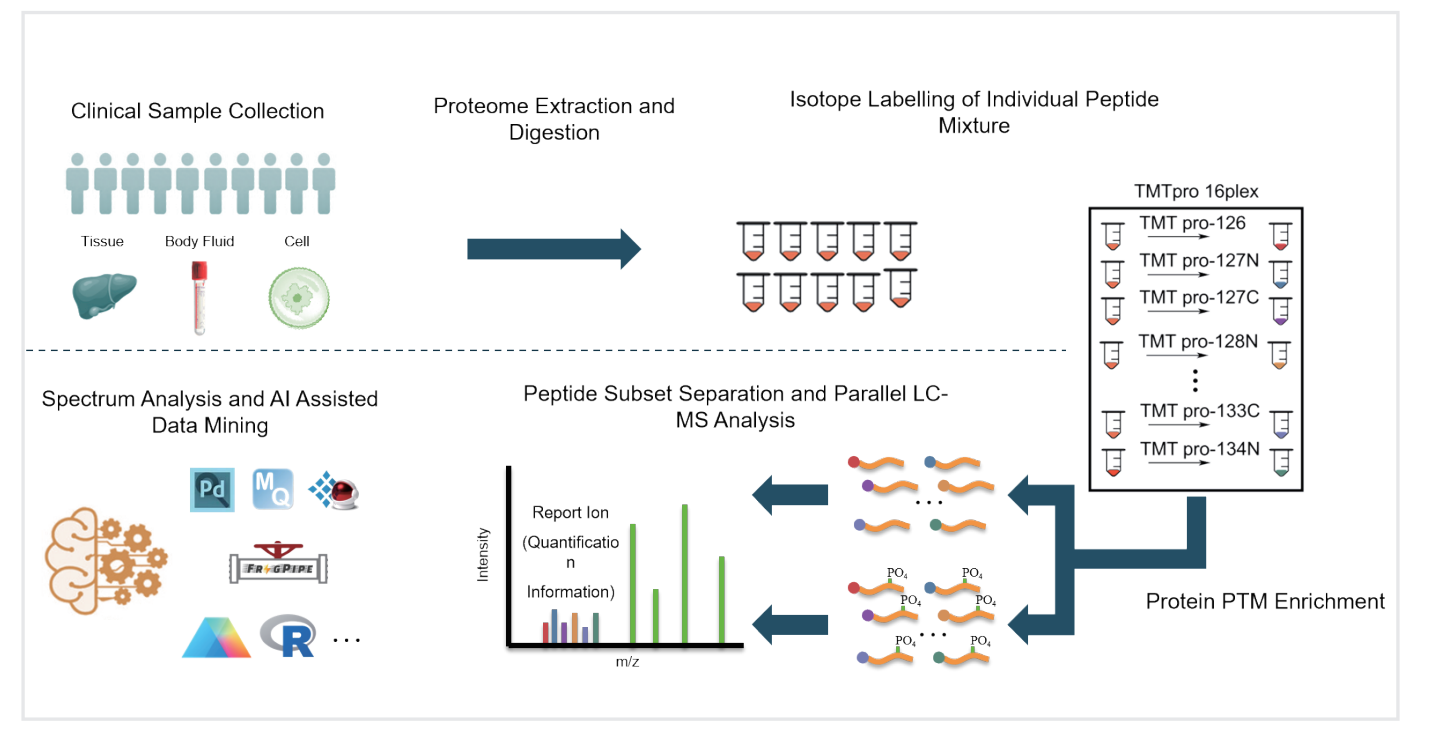 TMT-labeling proteomics and phosphoproteomics analysis
TMT-labeling proteomics and phosphoproteomics analysis
Mass Spectrometers
timsTOF Pro;
Bruker Orbitrap Exploris 480